artus HBV PCR Kits CE
应用real-time PCR定量检测乙型肝炎病毒DNA
应用real-time PCR定量检测乙型肝炎病毒DNA

Cat. No. / ID: 4506363
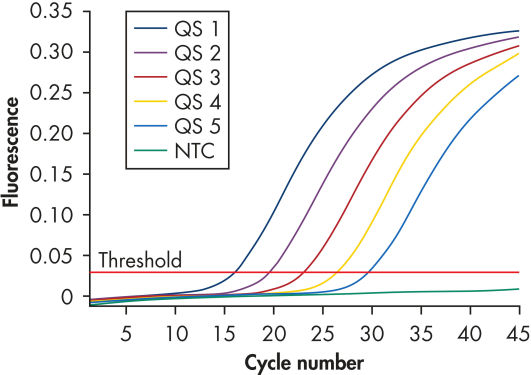
为确保高灵敏度,artus HBV Kits已经过优化,可检测到低拷贝数的乙型肝炎病毒DNA。artus HBV QS-RGQ Kit在QIAsymphony RGQ上使用的检测阈限为10.2 IU/ml(参见" Highly sensitive detection of HBV DNA")。(1 IU/ml相当于在Rotor-Gene Q实时荧光定量PCR分析仪上检测到8.21拷贝/毫升的乙型肝炎病毒DNA。转化因子为平均值。)
为确保高度特异性,已使用多种乙型肝炎病毒,包括A–H基因型和相关病原体对artus HBV Kits进行了验证。
| 试剂盒 | artus HBV RG PCR Kit | artus HBV QS-RGQ Kit |
|---|---|---|
| 经过验证的适用样本类型 | EDTA血浆 | EDTA血浆 |
| 检测阈限 | 3.8 IU/ml | 10.2 IU/ml |
| 线性范围 | 1.1至 >4 x 109 IU/ml | 31.6至 >2 x 107 IU/ml |
| 特异性 | 基因型为A–H的乙型肝炎病毒 | 基因型为A–H的乙型肝炎病毒 |
artus HBV Kits的原理是使用real-time PCR技术,扩增和检测乙型肝炎病毒基因组中一段特定序列。该试剂盒特异性强、灵敏度高(参见" Highly sensitive detection of HBV DNA"),且重复性好。每个artus HBV Kit都包含5个乙型肝炎病毒标准品(参见" Reliable quantitation of HBV load")。使用这些标准品可确保准确定量病毒。此外,该试剂盒还含有异源扩增系统,即内参,可识别PCR抑制剂。内参(IC)可在PCR中通过不同的荧光通道检测,而不会降低乙型肝炎病毒的检测灵敏度。
| 试剂盒 | artus HBV RG PCR Kit和artus HBV QS-RGQ Kit |
|---|---|
| 经验证的适用样本类型 | EDTA血浆 |
| 扩增子 | 乙型肝炎病毒基因组一段长为134 bp的核心序列 |
artus HBV PCR Kits包含对乙型肝炎病毒DNA进行可靠定量检测的所有所需试剂,并已经过优化。只需在即用型PCR预混液加入模板DNA(如使用LC试剂盒,需要在Mg溶液中也加入),即可按照试剂盒操作手册中的优化程序,在real-time PCR仪上开始反应。
QIAsymphony RGQ的乙型肝炎病毒检测解决方案包括:使用QIAsymphony SP进行样本制备,使用QIAsymphony AS进行反应体系构建,并在Rotor-Gene Q实时荧光定量PCR分析仪上使用artus HBV QS-RGQ Kit进行检测。这一体系不仅可可靠检测病原体,而且完全符合体外诊断CE要求(参见" Integrated QIAsymphony RGQ system for HBV detection")。
artus HBV LC、RG和TM PCR Kits已经过验证,适用于CE认证的QIAamp DSP Virus Kit从EDTA血浆中纯化得到的病毒RNA。
artus HBV RG、LC和TM PCR Kits可快速、灵敏的定量检测使用QIAamp DSP Virus Kit从人类血浆中纯化得到的乙型肝炎病毒DNA。这些试剂盒适用于ABI PRISM 7000、7700和7900HT SDS,LightCycler 1.1/1.2/1.5/2.0 Instruments或Rotor-Gene Q实时荧光定量PCR分析仪。
artus HBV QS-RGQ Kit适用于QIAsymphony RGQ仪器,整个工作流程覆盖样本制备到乙型肝炎病毒DNA定量检测,完全符合体外诊断CE要求。