QIAGEN Large-Construct Kit
ゲノムDNAフリーのBAC、PAC、P1(最高50 µg)、コスミドDNA(最高200 µg)の精製用
ゲノムDNAフリーのBAC、PAC、P1(最高50 µg)、コスミドDNA(最高200 µg)の精製用
✓ オンライン注文による24時間年中無休の自動処理システム
✓ 知識豊富で専門的な製品&テクニカルサポート
✓ 迅速で信頼性の高い(再)注文
Cat. No. / ID: 12462
✓ オンライン注文による24時間年中無休の自動処理システム
✓ 知識豊富で専門的な製品&テクニカルサポート
✓ 迅速で信頼性の高い(再)注文
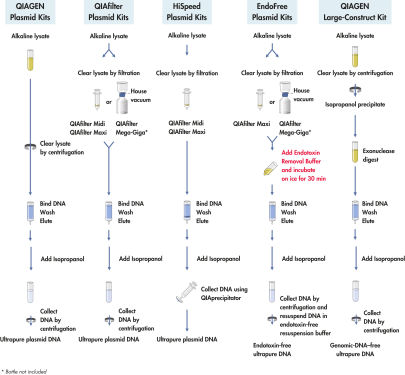
QIAGEN Large-Construct Kitはオープンカラム方式の陰イオン交換カラムで、高分子DNAの精製を行ないます。ユニークなATP依存性エキソヌクレアーゼ分解ステップが組み込まれているので、混入したゲノムDNAが選択的に除去されます。精製したDNAは、CsCl密度勾配遠心操作を2回行なって得られる精製グレードに匹敵し、トランスフェクション等のアプリケーションに最適です。
QIAGEN Large-Construct Kitを利用したDNA精製では、至適化されたオープンカラム調製法を利用し、汎用されている方法に比較して、有意に純度の高いDNAを精製可能です。ユニークなATP依存性エキソヌクレアーゼ処理が組み込まれているので、ゲノムDNAが効率的に除去されます。
QIAGEN Large-Construct Kitに含まれる、QIAGEN-tipの非常にユニークな陰イオン交換樹脂は核酸精製のみを目的として開発されました。本製品の優れた核酸分離能力により、CsCl密度勾配遠心操作を2回連続で行なって得たDNAの純度に匹敵、あるいはそれ以上の純度のDNAが調製されます。充填済みQIAGEN-tip(図" 陰イオン交換チップ")はオープンカラム方式を採用しており、プラスミド調製中にカラムが乾燥することがなく、作業者がカラムに付き添う必要がないので、実際の操作時間は短くてすみます。全てのQIAGENプラスミド精製システムでは、ユーザーおよび環境への影響が最小限となるように、フェノール、クロロホルム、臭化エチジウム、CsCl等の有害な試薬を一切使用していません。
500 mlまでの培養液のアルカリ溶解に続いて(フローチャート" QIAGEN Plasmid Kit操作手順")、本キットでは、ATP依存性エキソヌクレアーゼによるユニークな分解が組み込まれています。この製法により、切断、損傷したDNAおよび夾雑物としてのゲノムDNAの選択的な除去が確実になります。サンプルを陰イオン交換チップ上にロードすると、適切な低塩あるいはpH条件でプラスミドDNAが選択的に結合します。RNA、タンパク質、代謝物、その他の低分子量の不純物は中濃度の塩による洗浄で取り除かれます。ゲノムDNAフリーの高純度プラスミドDNAは高塩濃度バッファーで溶出されます。イソプロパノール沈殿によりDNAが濃縮および脱塩され、遠心操作により回収されます。
QIAGEN Large Construct Kitを用いて精製したDNAは以下のようなアプリケーションに最適です。
| Features | Specifications |
|---|---|
| Plasmid type | BAC, PAC, P1, cosmid DNA |
| Applications | Subcloning, transfection, sequencing etc. |
| Processing | Manual (centrifugation) |
| Culture volume/starting material | <500 ml culture volume |
| Samples per run (throughput) | 1 sample per run |
| Technology | Anion-exchange technology |
| Time per run or prep per run | 280 min |
| Yield | <150 ug |