New Rotor-Gene AssayManager v2.1 software for QIAGEN CE-IVD assays only.
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Rotor-Gene AssayManager v2.1
Cat. No. / ID: 9024203
✓ 24/7 automatic processing of online orders
✓ Knowledgeable and professional Product & Technical Support
✓ Fast and reliable (re)-ordering
Features
- Standardized, fully automated real-time PCR analysis
- Maximized process safety and ease of use
- Unprecedented flexibility for all routine testing demands
- CE-IVD compliant QIAGEN assays only
- Enhanced lab productivity with optimized workflows
Product Details
IMPORTANT:
- Rotor-Gene AssayManager v2.1 is a unique software platform and it is not an update for Rotor-Gene AssayManager v1.0
- Rotor-Gene AssayManager v2.1 requires distinct plug-ins that are not compatible with Rotor-Gene AssayManager v1.0
- Rotor-Gene AssayManager v2.1 is not compatible with Rotor-Gene AssayManager v1.0 plug-ins
- Rotor-Gene AssayManager v2.1 cannot run on the same computer as Rotor-Gene AssayManager v1.0 software
Rotor-Gene AssayManager v2.1 software is used in combination with Rotor-Gene Q and Rotor-Gene Q MDx instruments for routine testing. This intuitive software manages all tasks associated with running real-time PCR assays offering enhanced convenience, standardization, and flexibility. Fully automated analysis and interpretation of results minimizes human error, delivering results you can trust. With a range of kits for a CE-IVD compliant workflow, Rotor-Gene AssayManager v2.1 covers the demands of routine testing laboratories.
Principle
Rotor-Gene AssayManager v2.1 enables:
- Operation of up to 4 Rotor-Gene Q or Rotor-Gene Q MDx cyclers from one computer
- Individual or network installation with remote access
- IVD-compliant data management
- IVD-compliant user management
- Fully automated analysis and interpretation of results
- Validated workflows for QIAGEN CE-IVD assays
Procedure
Comprehensive user guidance with flexible options
Rotor-Gene AssayManager v2.1 operates in a single Closed Mode. Color schemes, easy-to-understand icons, and comprehensive help and warning messages guide the user step-by-step through the software.
Control of up to 4 Rotor-Gene Q cyclers and remote access
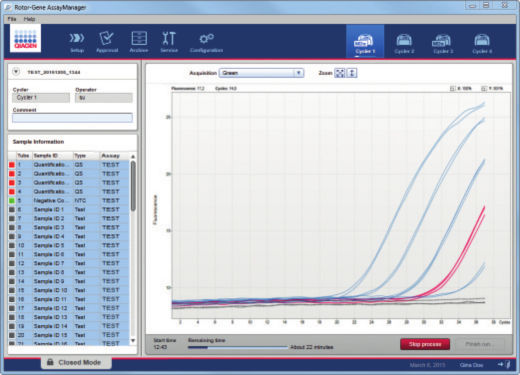
Rotor-Gene AssayManager v2.1 controls the Rotor-Gene Q or Rotor-Gene Q MDx cycler, i.e., the software will provide all functions to set up, start and run real-time PCR experiments on up to 4 Rotor-Gene Q or Rotor-Gene Q MDx cyclers in parallel (see figure " Increased lab efficiency"). This saves laboratory space and increases lab efficiency.
Rotor-Gene AssayManager v2.1 can also be used for experiment setup, result approval or reporting on computers not connected to a Rotor-Gene Q or Rotor-Gene Q MDx cycler. In this case, the software can be installed on the user's office computer enabling convenient result review and approval. Alternatively, a computer in the assay setup room may be used for run setup. This networking capability together with secure database management simplifies a variety of everyday tasks.
Fully automated real-time PCR analysis
Automated real-time PCR analysis is the core function of Rotor-Gene AssayManager v2.1. Rotor-Gene AssayManager analyzes the real-time PCR raw data according to well-defined assay specific rules and generates result reports comprising information on the validity or invalidity of the assay and individual samples.
For QIAGEN validated assays, the unique Automatic Data Scan (AUDAS) compares real-time data with performance characteristics established by statistical methods during assay development. This feature detects assay deviations and artifacts that cannot be monitored by the assay controls alone, ensuring best-in-class, error-free real-time PCR analysis.
Reliable data management
All data from all installations and from all Rotor-Gene Q or Rotor-Gene Q MDx cyclers in your lab are stored in a central database. After analysis, approval and reporting (e.g., as a printout, PDF or LIMS export), data is moved to a common electronic archive. For fast and easy data access of all data at each time point, the database is searchable by using various filter functions.
Secure user management
Different user profiles are available and are password protected (user login is required) for increased security. Operators set up and start a run, while administrators are responsible for managing all assets (e.g., cyclers, assay profiles, users) necessary for working with Rotor-Gene AssayManager v2.1. Approvers accept or reject the results of a run and release the data of an experiment. Assignment of these dedicated user roles enables optimal use of resources for your laboratory tasks.
Support of 21 CFR Part 11 compliance
Rotor-Gene AssayManager v2.1 includes specific features that support the technical requirements of 21 CFR Part 11 regulations, enabling use of an electronic records system. User accounts are password-protected, including special security requirements, and all user actions are documented in an audit trail that can be filtered and printed out. The Rotor-Gene AssayManager 2.1 audit trail is designed based on guidelines in FDA CFR Title 21, Part 11 Electronic Records, Electronic Signatures.
Software that meets the demands of routine testing applications — today and tomorrow
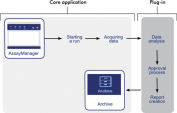
Rotor-Gene AssayManager v2.1 standardizes procedures such as cycler control and data storage, helping to reduce the burden of multiple standard operating procedures and simplifying laboratory documentation (see figure " Standardized workflow"). Full flexibility is retained through the use of modular plug-ins that ensure future assays and different assay designs can be run with the workflow already established in your laboratory.
See figures
Applications
Software
To download Rotor-Gene AssayManager v2.1 after purchase:
- Click the "Download" button and follow the on-screen instructions to install Rotor-Gene AssayManager v2.1.
Rotor-Gene AssayManager v2.1 and license must be purchased prior to use.
IMPORTANT: In order to perform and analyze runs, at least one Rotor-Gene AssayManager v2.1 plug-in needs to be installed. Refer to your assay kit handbook for more information.
To purchase a license key for Rotor-Gene AssayManager v2.1:
- 1. After purchasing Rotor-Gene AssayManager v2.1 software and licenses, click the "Download" button and follow the onscreen instructions to install. After successful installation of the Rotor-Gene AssayManager v2.1 and a plug-in, a “key file” will be generated. This "key file" will be attached to the "Technical Support Form" in Step 5, below.
2. Go to the Technical Support Form. Make sure to include all required information and your Rotor-Gene Q or Rotor-Gene Q MDx serial number when filling out the form.
3. For Question 2, "Type of Request", select “Other”.
4. For Question 4, “Your inquiry”, enter "Rotor-Gene AssayManager license request” and include your purchase order number, from your order confirmation.
5. Include the "key file" from Step 1 as an attachment.
6. Submit the form. QIAGEN Technical Services will respond via email with a license file within 1–2 working days.
7. After receiving your computer-specific Rotor-Gene AssayManager v2.1 license file, archive the license file locally on your computer.
Supporting data and figures
Increased lab efficiency.